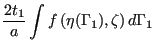 |
A Möhring element is a vortex element for which the characteristics are determined by the integration of a nonlinear differential equation describing the physics of the problem [52]. It basically describes the flow in narrow gaps between a rotating and a static device and is more precise than the formulation of the forced and free vortex element. The geometry is shown in Figure 100 and consists of a minimum radius, a maximum radius, a value for the gap between stator and rotor and the shroud radius. It is complemented by the label of the upstream and downstream node, the rotating speed of the rotor and the value of the swirl at entrance. The user must choose the centrifugal or centripetal version of the Moehring element before start of the calculation, i.e. the user must decide beforehand in which direction the flow will move. If the calculation detects that the flow is reversed, an error message is issued.
The following constants must be entered (to be specified in that order on the line beneath the *FLUID SECTION, TYPE=MOEHRING CENTRIFUGAL card or *FLUID SECTION, TYPE=MOEHRING CENTRIPETAL card):
Example files: moehring.